LBL-01791 [B] Laborie alpHaONE User & Service Manual 77
Appendix A: Safety Information
ThesafetyinformationinthisAppendixisintendedforthefollowingusers:
•
Those performing investigations by using the alpHaONE measurement system
•
Those who install, test, and maintain the alpHaONE measurement system
BeforeworkingwiththealpHaONEmeasurementsystemandthismanual,pleasetakenoticeofthesafetyinformation
as described in this appendix.
The alpHaONE measurement system is a prescription device to be used only by licensed and trained healthcare
professionals or individuals who have been trained and authorized by a healthcare professional or by a medical
institution, under the supervision of a healthcare professional. The alpHaONE system must be purchased under the
supervision of a healthcare professional.
The contents of this manual should not be considered to be all inclusive or covering all contingencies. Please direct
any questions on the use of this product or interpretation of data acquired from the product to the responsible
healthcare professional(s) at your institution.
For U.S. only: Caution: Federal law restricts this device to sale by or on the order of a licensed and trained healthcare
professional.
ThealpHaONEmeasurementsystemcomplieswiththerequirementsasstatedbyIEC60601-13rdedition(Medical
ElectricalEquipment),ANSI:AAMIES60601-1:2005,CAN/CSAC22.2NO.60601-1:2014,IEC60601-1-2:2014(EMC)
andtheMedicalDeviceDirective(93/42/EEC).
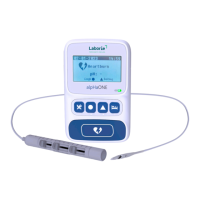
pH Capsule
•
Classiedbyelectricshockprotectiontype:devicefedbyinternalpowersupply.
•
Classiedbyelectricshockprotectionlevel:BF.
•
Classiedbyliquidprotectionlevel:IPX8.
•
Classiedbysafetylevelofuseinthepresenceofinammableanaestheticgasmixedwithairorwithoxygen
ornitrousgas:devicecannotbeusedinthepresenceofinammableanaestheticgasmixedwithairorwith
oxygen or nitrous gas.
•
Classiedbyoperatingmode:continuousoperation.
 Loading...
Loading...