8 Laborie alpHaONE User & Service Manual LBL-01791 [B]
Software and Computer
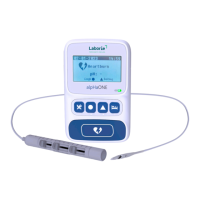
The alpHaONE system is used in conjunction with a computer and the Laborie alpHaONE application. To run the
application, the computer must be running Windows 10 Professional Edition (64 bit).
1
CAUTION
For U.S. only: Federal law restricts this device to sale by or on the order of a trained and licensed healthcare
professional.
The alpHaONE system connects to the computer via the supplied USB interface cable.
Hardware and Software Changes
WhenyoureceivenewalpHaONEmanuals,alwayskeeptheoriginalmanualsassuppliedwithyoursystem,as
descriptions and images may differ as a result of chages and upgrades to the alpHaONE hardware and software.
Safety Information
The alpHaONE User’s & Service Manual is intended for all users of the alpHaONE measurement system. Before
workingwiththesystemanditsmanual,carefullyreadthesafetyinformationpresentedinAppendixA.
Clinical Procedures
AllowonlythosehealthcareprofessionalswhohavereceivedadequatetrainingabouttheoperationofthepHCapsule
Monitoring System to use this product. Clinical judgements and protocols are left to the discretion of the individual
healthcare professional.
Risks
Extensive clinical trials suggest that the pH Capsule procedure is generally safe and tolerable for patients. However,
there might be conditions in which some patients can experience adverse effects or discomfort. Possible adverse
effects of endoscopic gastrointestinal tract examination include, but are not limited to, perforation, hemorrhage,
aspiration, fever, infection, high blood pressure, breath arrest, arrhythmia, and cardiac arrest.
Possible adverse effects of alpHaONE pH examination include, but are not limited to, the following:
•
Failure to detach and release capsule from the delivery system.
•
Capsule detaches early.
•
Capsule failed to detach from esophagus within several days after placement. Foreign body granuloma may be
formed if the capsule detachment is delayed.
•
Discomfort caused by the placement of the capsule, such as chest pain, odynophagia, and foreign body
sensation.
•
Hemorrhage caused by esophageal mucosal or submucosal tear, which requires immediate treatment.
•
Perforation.
•
Possible side effects such as sore throat, throat discomfort, and soft-tissue injury.
IMPORTANT
If the capsule fails to detach and release from the delivery system, refer to AppendixB,“Handling
ProcedurefortheDeliverySystem” on page 82 for guidance on how to resolve the problem.
1 Windows10isatrademarkofMicrosoftCorporation,USA.TheMicrosoftOceUserInterfaceissubjecttoprotectionunder
U.S. and international intellectual property laws and is used by Laborie Medical Technologies Corp. under license from Microsoft.
 Loading...
Loading...