Basic Chemistry and Basic Corrosion Theory 2:2
CP 1 – Cathodic Protection Tester Course Manual
© NACE International, 2000
02/01/05
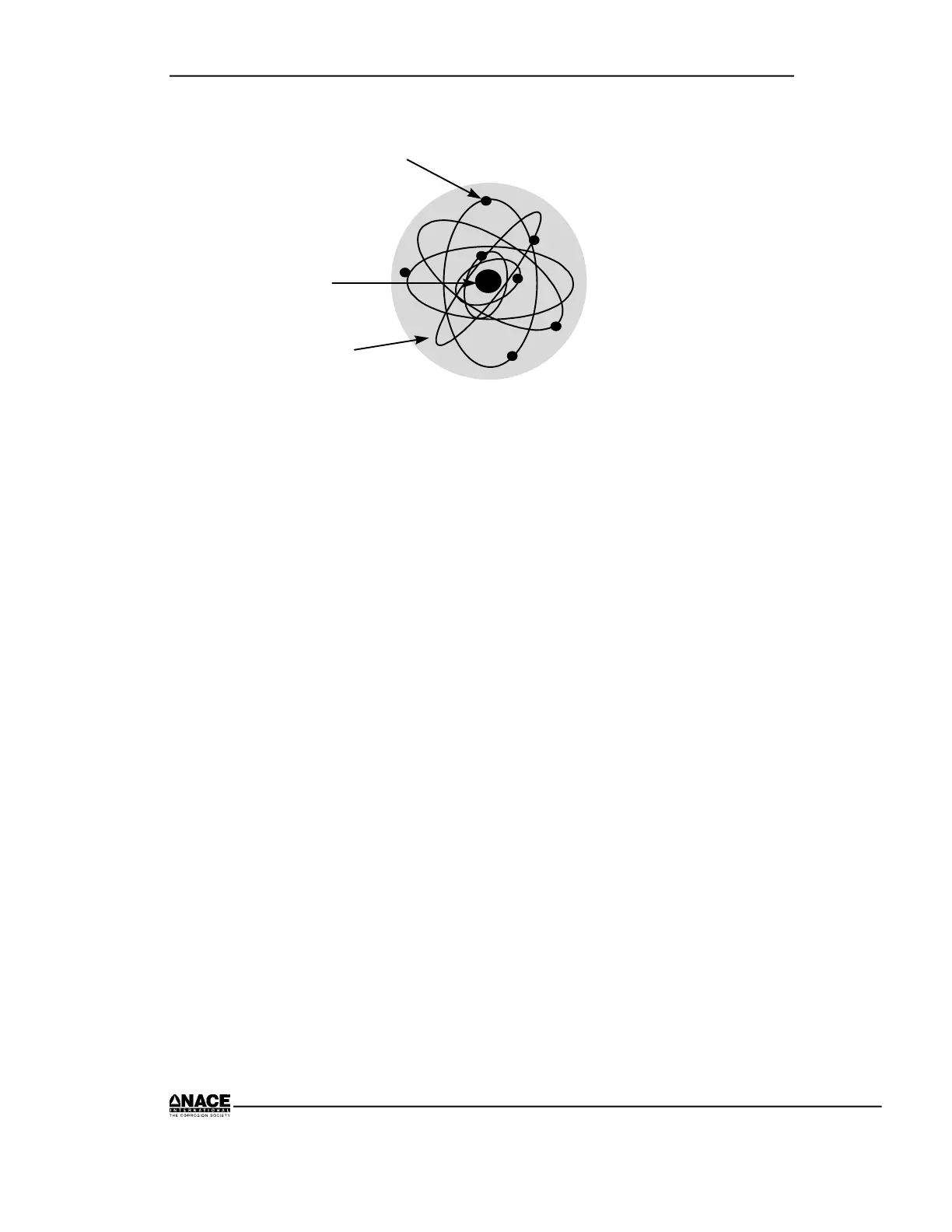
ELECTRONS
UCLEUS
ORBIT
Figure 2.1 The Bohr Model of an Atom
Ions
Gaining or losing electrons can electrically charge atoms. Electrically
charged atoms are referred to as ions and the charge on an atom is known as
its valence state. An ion formed by the gaining of electrons is called an
anion and is negatively charged because the number of electrons is greater
than the number of protons. The loss of electrons yields a positively
charged ion called a cation. For example, when sodium combines with
chlorine an electron is transferred from sodium to chlorine, creating a
positively charged sodium ion and a negatively charged chloride ion. The
two ions, now of opposite charge, are electrostatically attracted to each
other, forming a molecule. Ions allow for transfer of electrical charge in
liquids.
Compounds (Molecules)
Molecules are composed of two or more atoms. A molecule is the smallest
unit of a substance with the same specific chemical properties of that
substance. For example, a single water molecule is composed of one oxygen
and two hydrogen atoms as shown in Figure 2.2. Further splitting of this
molecule would result in a substance with characteristics unlike water. The
atoms of a molecule are held together by a force referred to as chemical
bonding. It is this chemical bonding that defines many of the properties of a
substance.
 Loading...
Loading...