Basic Chemistry and Basic Corrosion Theory 2:9
CP 1 – Cathodic Protection Tester Course Manual
© NACE International, 2000
02/01/05
Corrosion Cell
Corrosion is an electrochemical process involving the flow of electrons and
ions. Metal loss (corrosion) occurs at the anode. No metal loss occurs at the
cathode (the cathode is protected).
Electrochemical corrosion involves the transfer of electrons across
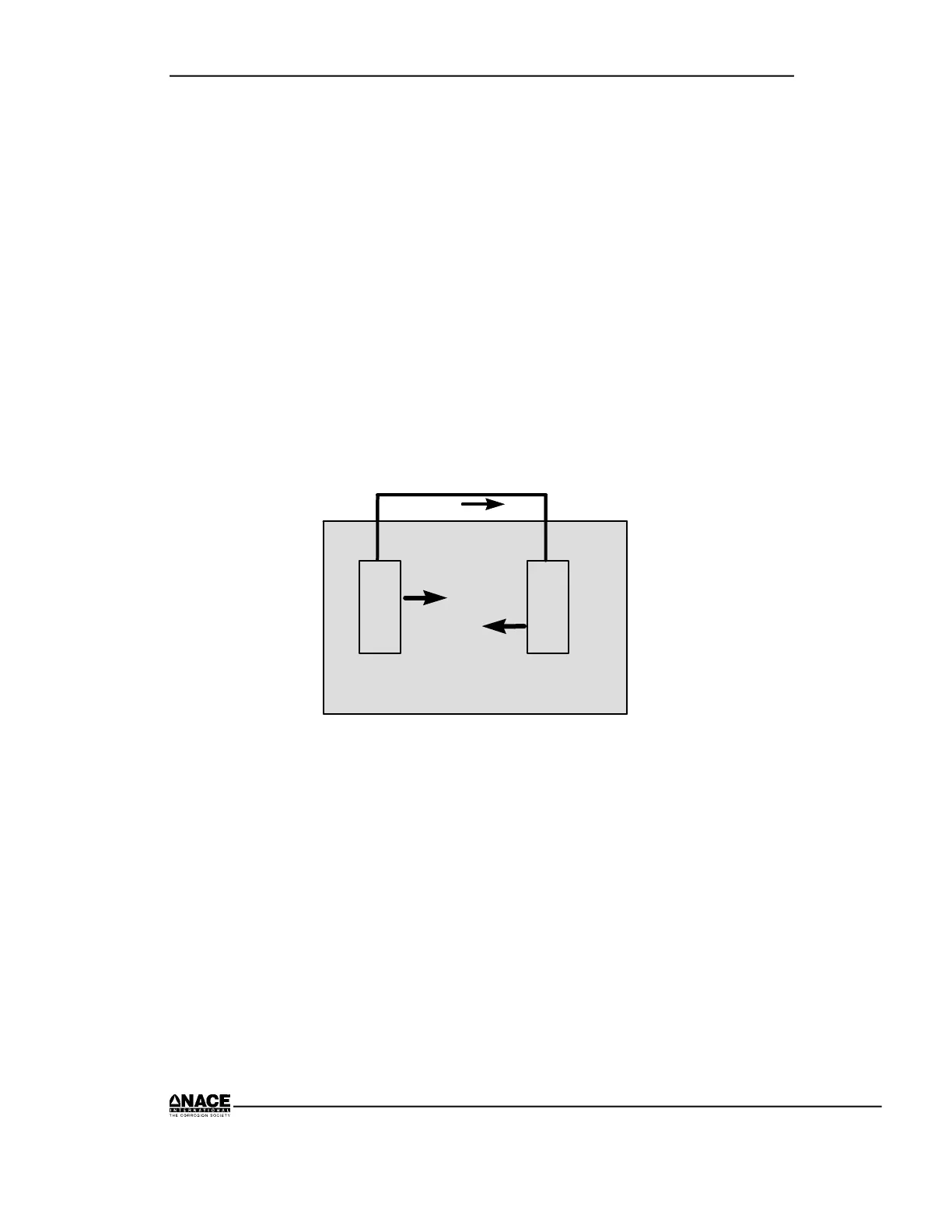
metal/electrolyte interfaces. Corrosion occurs within a corrosion cell. A
corrosion cell consists of four parts as illustrated in Figure 2.8.
• Anode
• Cathode
• Electrolyte
• Metallic Path
Anode
e-
Metallic Path
+ ions
-ions
Electrolyte
Cathode
Anode
e-
Metallic Path
+ ions
-ions
Electrolyte
Cathode
Figure 2.8 Corrosion Cell
Anode Reactions
The chemical reaction that occurs at the anode, the anodic reaction, is an
oxidation reaction. Corrosion is the result of the oxidation reaction in a
corrosion cell. Oxidation is the loss of electrons as shown in the following
reaction:
M
o
→ M
n+
+ ne
-
where n is the number of electrons involved.
 Loading...
Loading...