Safety Information
T3300 Diagnostic Ultrasound System | 13 | B00601-010 4/10/17
Guidance and Manufacturer’s Declaration - Electromagnetic Immunity - Non-life-supporting
Equipment
Voltage dips/
Dropout IEC
61000-4-11
>95% dip for 0.5 cycle
60% dip for 5 cycles
30% dip for 25 cycles
>95% dip for 5 seconds
As specified
Mains power quality should be
that of a typical commercial or
hospital environment. If the user
of the T3300 Diagnostic
Ultrasound System requires
continued operation during
power mains interruptions, it is
recommended that the T3300
Diagnostic Ultrasound System
be powered from an
uninterruptible power supply or
battery.
Power
frequency 50/
60 Hz
Magnetic field
IEC 61000-4-8
3 A/m As specified
Power frequency magnetic
fields should be that of a typical
commercial or hospital
environment.
Table 6 Guidance and Manufacturer’s Declaration - Electromagnetic Immunity - Non-life-supporting
Equipment
Immunity test IEC 60601 test level Compliance level
Electromagnetic environment -
Guidance
Portable and mobile RF
communications equipment
should be used no closer to any
part of the T3300 Diagnostic
Ultrasound System, including
cables, than the recommended
separation distance calculated
from the equation applicable to
the frequency of the transmitter.
Recommended separation
distance
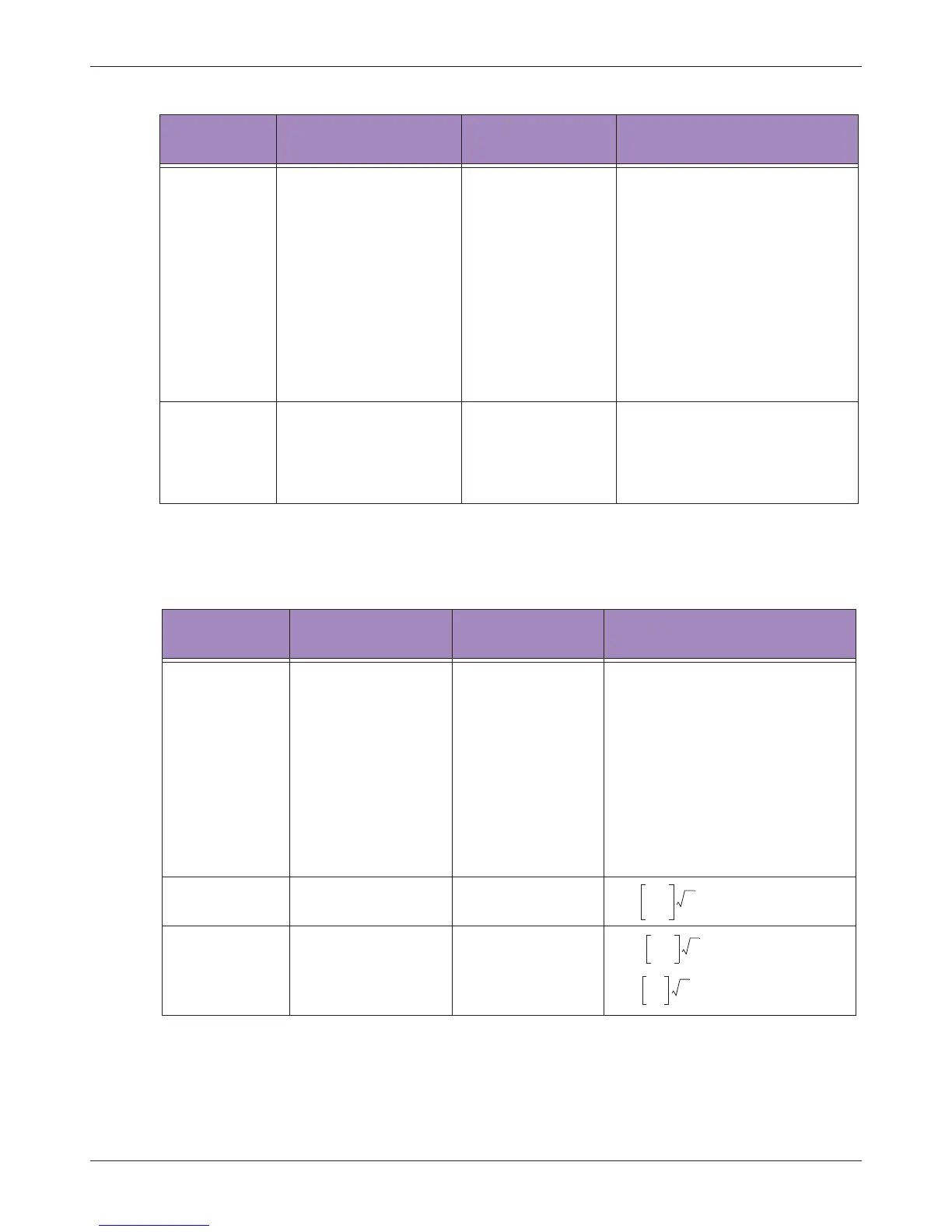
Conducted RF
IEC 61000-4-6
3 Vrms
150 KHz to 80 MHz
[V1] = 3 Vrms
Radiated RF
IEC 61000-4-3
3 V/m
80 MHz to 2.5 GHz
[E1] = 3 V/m
80 MHz to 800 MHz
800 MHz to 2.5 GHz
Table 5 Guidance and Manufacturer’s Declaration - Electromagnetic Immunity - All Equipment
Immunity test IEC 60601 test level Compliance level
Electromagnetic environment
- Guidance
 Loading...
Loading...