P/N MAN-01964 Revision 001 7
DRAFT
Preview copy—Generated 7/9/2010
User Manual
Chapter 1—General Information
Compliance
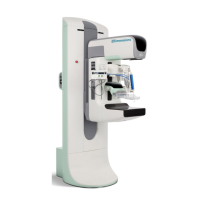
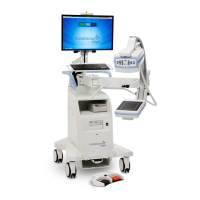
3.0 Compliance
This section describes the mammography system compliance requirements and the
responsibilities of the manufacturer.
3.1 Compliance Requirements
The manufacturer has the responsibility for the safety, reliability, and performance of this
equipment with the following provisions:
• The electrical installation of the room meets all requirements.
• The equipment is used according to Instructions for Use.
• The assembly operations, extensions, adjustments, changes, or repairs are performed
only by authorized persons.
• The network and communication equipment must be installed to meet IEC Standards.
The complete system (network and communications equipment and Selenia Dimensions
Mammography System) must be in compliance with IEC 60601-1 and IEC 60601-1-1.
3.2 Compliance Statements
The manufacturer states this device is made to meet the following requirements:
• CAN/CSA ISO 13485:2003
• EN 60601-1:1990 +A1+A11+A12+A2+A13 Medical Electrical Equipment—General
Requirements for Basic Safety and Essential Performance
• FDA, 21 CFR [Parts 820, 900 and 1020]
• IEC 60601-1:1988 +A1+A2:1995 +A13:1996 Medical Electrical Equipment—General
Requirements for Safety
• IEC 60601-1-1:2000-12 Medical Electrical Equipment—Collateral Standard: Safety
Requirements for Medical Electrical Systems
• IEC 60601-1-2:2001 Medical Electrical Equipment—Collateral Standard:
Electromagnetic Compatibility for Medical Electric Systems
• IEC 60601-1-3:1994 Medical Electrical Equipment—Collateral Standard: Requirements
for Radiation Protection in Diagnostic X-ray Equipment
• IEC 60601-1-4:1996 +A1:1999 Medical Electrical Equipment—Collateral Standard:
Programmable Electrical Medical Systems
• IEC 60601-2-7:1998 Medical Electrical Equipment—Particular Requirements for the
Safety of High-Voltage Generators of Diagnostic X-ray Equipment
• IEC 60601-2-28:1993-03 Medical Electrical Equipment—Particular Requirements for the
Safety of X-ray Source Assemblies and X-ray Tube Assemblies for Medical Diagnosis
• IEC 60601-2-32:1994 Medical Electrical Equipment—Particular Requirements for the
Safety of Associated Equipment of X-ray Equipment
• IEC 60601-2-45:2001 Medical Electrical Equipment—Particular Requirements for the
Safety of Mammographic X-ray Equipment and Mammographic Stereotactic Devices
• UL 60601-1: Medical Electrical Equipment, Part 1—General Requirements for Safety
• CAN/CSA: Medical Electrical Equipment Part 1: C22.2 No. 601.1–M90—General
Requirements for Safety
 Loading...
Loading...