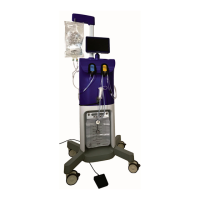
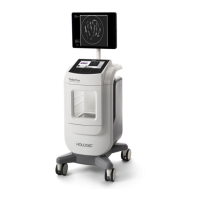
Fluent Pro Fluid Management System
English Introduction 3
• Ensure the patient’s body temperature is monitored throughout the entire surgery. Continuous flow of
distention fluids during hysteroscopic surgery can lead to a lowering of the patient’s body temperature.
Low body temperatures can cause coronary and cardiovascular problems. Longer operating times and
the use of cold distension media should be avoided.
• For your own safety and that of your patient, use only Fluent Pro accessories referenced in Table 9:
Disposables and Accessories.
• The system is not explosion-proof. Do not use in an area where flammable anesthetic gases are present.
• If a system defect is suspected or confirmed, do not use the system. Ensure the system is fully functional
as described in Section 6.1, Annual Inspection.
• Fluent Pro Procedure Kits are single use disposables. Do not re-use or reprocess any of the components
contained within the single-use Procedure Kit.
1.2.2.2 List of Precautions
Caution!
• Do not sterilize or immerse the Fluent Pro Fluid Management System in disinfectant.
• Electrical safety testing should be performed by a biomedical engineer or other qualified person.
• The Fluent Pro Fluid Management System requires special precautions regarding electromagnetic safety
and needs to be installed and put into service according to the electromagnetic safety information
provided in this manual.
• Portable and mobile RF communications equipment, including cellular telephones and other wireless
devices, can affect medical electrical equipment. To avoid damage to or malfunction of the system, do
not operate communications equipment or cellular telephones at a distance closer than specified in
Section 6.4 Electromagnetic Compatibility.
• Use of the Fluent Pro Fluid Management System adjacent to or stacked with other equipment should be
avoided because it could result in improper operation. If such use is necessary, the Fluent Pro Fluid
Management System and the other equipment should be observed for proper operation before
performing a procedure.
• This equipment is designed and tested to minimize interference with other electrical equipment.
However, if interference occurs with other equipment, it may be corrected by one or more of the
following measures:
o Reorient or relocate this equipment, the other equipment, or both.
o Increase the separation between the pieces of equipment.
o Connect the pieces of equipment into different outlets or circuits.
o Consult a biomedical engineer.
1.3
System Introduction
1.3.1 Introduction to the Fluent Pro Fluid Management System
The Fluent Pro Fluid Management System is designed to provide liquid distension of the uterus during diagnostic and
operative hysteroscopy while monitoring the volume differentials between fluid flowing into and out of the uterus.
Additionally, the Fluent Pro Fluid Management System supports the use of the MyoSure
®
tissue removal devices for tissue
removal.
1.3.1.1 Technical Application Scope of the System
The Fluent Pro Fluid Management System allows Intrauterine Pressure to be adjusted between 40 and 150
mmHg. The system inflow rates will automatically adjust to reach and maintain the preset Intrauterine
Pressure setting while the system outflow rates are adjusted by the user through preset suction settings. The
system has been designed to provide both fluid and vacuum systems that maximize the performance of the
MyoSure Tissue Removal Device. The system provides varying levels of fluid suction.
1.3.1.2 Suggested Distension Media
The Fluent Pro Fluid Management System should only be used with sterile media. The Fluent Pro Fluid
Management System can be used with hypotonic, electrolyte-free media (e.g., glycine 1.5% and sorbitol
3.0%) and isotonic, electrolyte containing media (e.g., saline 0.9% and Lactated Ringer’s). See “List of
Warnings,” Section 1.2.2.1, for risks related to viscosity and the use of a high viscosity media such as Hyskon.