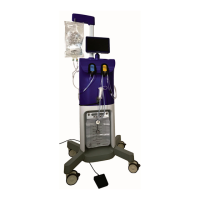
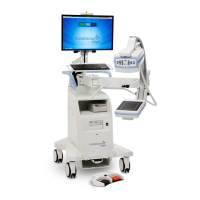
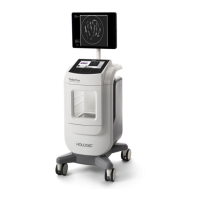
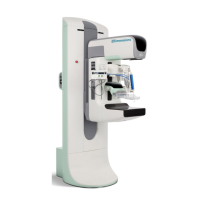
Fluent Pro Fluid Management System
82 Supplementary Information English
6.9.4 Complaints Reporting:
Report any complaints or problems with the quality, reliability, safety, or performance of this product to Hologic. If the
device has caused or added to patient injury, immediately report the incident to Hologic Authorized Representative and
Competent authority of the respective member state or country. The Competent Authorities for medical devices are usually
the individual Member States’ Ministry of Health, or an agency within the Ministry of Health.
6.9.5
Contacting Hologic Technical Support
Contact Hologic Technical Support to arrange for proper disposal of the Fluent Pro Fluid Management System in
accordance with the WEEE Directive.
Hologic Technical Support
United States and Canada:
Hologic, Inc
250 Campus Drive
Marlborough, MA 01752
Phone: 1.800.442.9892 (toll-free)
www.hologic.com
6.10
Symbol Glossary
Table 10: Symbols
Symbol
Standard Reference
& Symbol Number
Title of Symbol Description of Symbol Symbol
Standard Reference
& Symbol Number
Title of Symbol Description of Symbol
EN ISO 15223-1 5.1.1
ISO 7000-3082
Manufacturer
Indicates the medical device
manufacturer
EN ISO 15223-1 5.1.5
ISO 7000- 2492
Batch code
Indicates the manufacturer’s
batch code so that the batch
or lot can be identified
EN ISO 15223-1 5.1.3
ISO 7000-2497
Date of
manufacturer
Indicates the date when the
medical device was
manufactured
EN ISO 15223-1 5.1.6
ISO 7000- 2493
Catalog Number
Indicates the manufacturer’s
catalogue number so that the
medical device can be
identified
EN ISO 15223-1 5.1.11
ISO 7000-6049
Country of
manufacture
To identify the country of
manufacture of products
EN ISO 15223-1 5.1.7
ISO 7000-2498
Serial Number
Indicates the manufacturer’s
serial number so that a
specific medical device can
be identified
EN ISO 15223-1 5.1.4
ISO 7000-2607
Use-by date
Indicates the date after which
the medical device is not to
be used
EN ISO 15223-1 5.2.3
ISO 7000-2501
Sterilized using
ethylene oxide
Indicates a medical device
that has been sterilized using
ethylene oxide
EN ISO 15223-1 5.2.6
ISO 7000-2608
Do not resterilize
Indicates a medical device
that is not to be resterilized
EN ISO 15223-1 5.2.11
ISO 7000-3707
Single sterile
barrier system
Indicates a single sterile
barrier system
EN ISO 15223-1 5.4.2
ISO 7000-1051
IEC 60601-1,
Table D.1, 28
Do not re-use
Indicates a medical device
that is intended for one single
use only
EN ISO 15223-1 5.3.8
ISO 7000-2620
Humidity
limitation
Indicates the range of
humidity to which the
medical device can be safely
exposed
EN ISO 15223-1 5.2.8
ISO 7000-3082
Do not use if
package is
damaged and
consult
instructions for
use
Indicates that a medical
device that should not be
used if the package has been
damaged or opened and that
the user should consult the
instructions for use for
additional information
EN ISO 15223-1 5.3.9
ISO 7000-2321
Atmospheric
pressure
limitation
Indicates the range of
atmospheric pressure to
which the medical device can
be safely exposed
EN ISO 15223-1 5.3.7
ISO 7000-0632
Temperature
limit
Indicates the temperature
limits to which the medical
device can be safely exposed
EN ISO 15223-1 5.7.7 Medical Device
Indicates the item is a
medical device
EN ISO 15223-1 5.4.3
ISO 7000-1641
IEC 60601-1,
Table D.1, 11
Consult
instructions for
use
Indicates the need for the
user to consult instructions
for use
EN ISO 15223-1 5.7.10
Unique device
identifier
Indicates a carrier that
contains unique device
identifier information