Fluent Pro Fluid Management System
English Supplementary Information 83
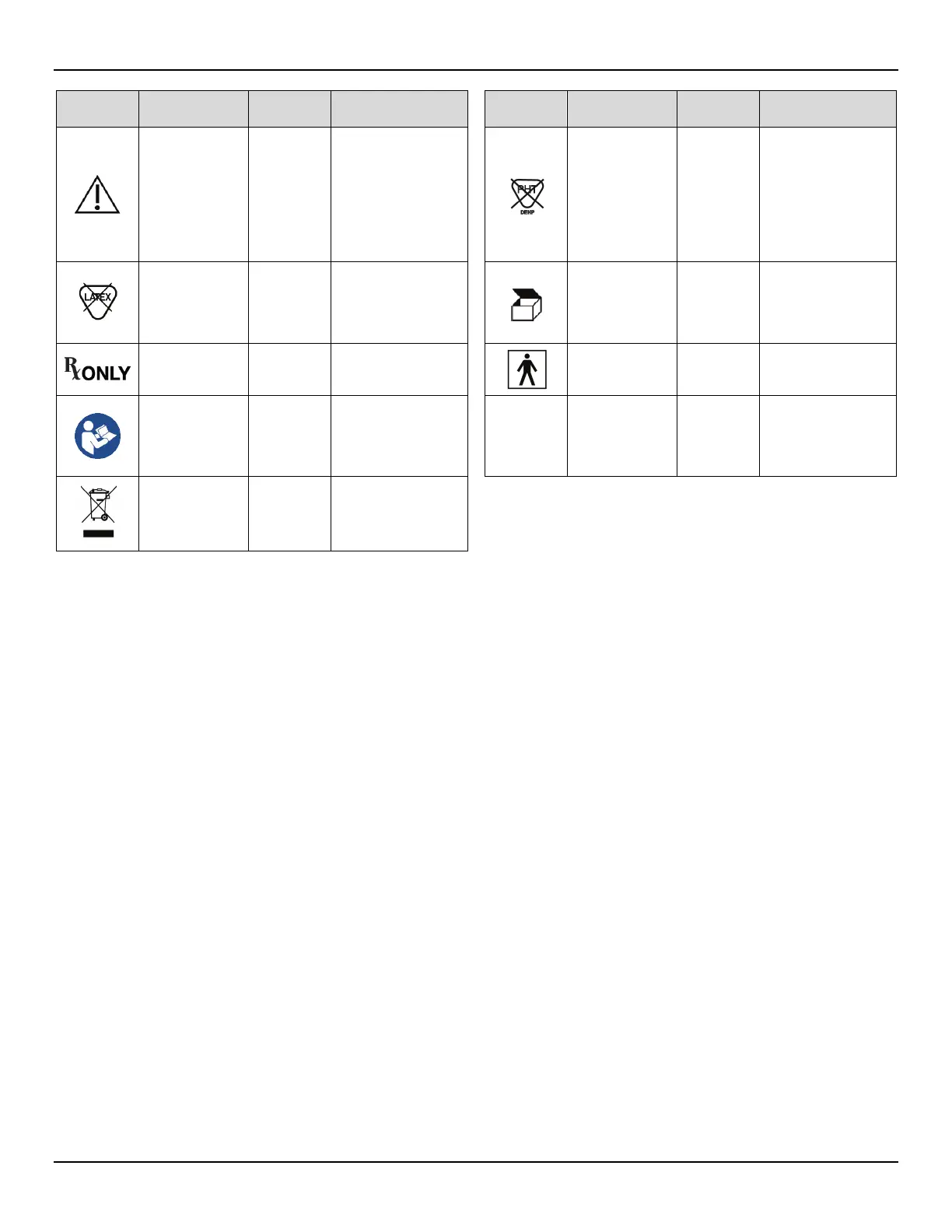
Symbol
Standard Reference
& Symbol Number
Title of Symbol Description of Symbol Symbol
Standard Reference
& Symbol Number
Title of Symbol Description of Symbol
EN ISO 15223-1 5.4.4
ISO 7000- 0434A
Caution
Indicates that caution is
necessary when operating
the device or control close to
where the symbol is placed,
or that the current situation
needs operator awareness or
operator action in order to
avoid undesirable
consequences
EN 15986 4.2, Annex A
and Annex B
ISO 7000-2725
phthalates
(DEHP)
Indicates the item does not
contain phthalates (DEHP)
EN ISO 15223-1 5.4.5
and Annex B
ISO 7000-2725
Product is not
made from
natural rubber
latex
Indicates that natural rubber
latex was not used in the
manufacturing of the
product, its container, or its
packaging.
ISO 7000-2794 Packaging unit
To indicate the number of
pieces in the package.
FDA 21 CFR 801.109
Prescription use
only
Caution: Federal law restricts
this device to sale by or on
the order of a physician
IEC 60417-5333
Type BF applied
part
Identifies a type BF applied
part complying with
IEC 60601-1
ISO 7010-M002 IEC 60601-1
Refer to instruction manual/
booklet.
To signify that the instruction
manual/booklet must be
read.
IP21
IEC 60529
IEC 60601-1
Degrees of
protection
provided by
enclosures
System is protected against
solid objects of 12.5mm or
greater and protected against
vertically falling drops of
water condensation.
IEC 60417-6414
EN 50419
Directive 2012/19/EU
WEEE; Waste
Electrical and
Electronic
Equipment
To indicate that separate
collection for waste electric
and electronic equipment
(WEEE) is required.
Hologic, Fluent, MyoSure, In-FloPak, Out-FloPak and associated logos are registered trademarks of Hologic, Inc. and/or its subsidiaries in the
United States and other countries. All other trademarks, registered trademarks, and product names are the property of their respective owners.
©2018-2024 Hologic Inc.
 Loading...
Loading...