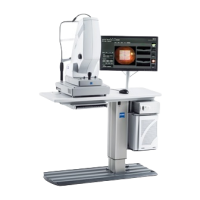
Instructions for Use 2 Introduction
2660021169042 Rev. A 2018-03 2.1 Scope
2660021169042 Rev. A 2018-03 29 / 246
2 Introduction
2.1 Scope
2.1.1 Intended Use
The PLEX® Elite 9000, a Swept-Source OCT [SS-OCT], is a non-
contact, high resolution, wide field-of-view tomographic and
biomicroscopic imaging device intended for in-vivo viewing, axial
cross-sectional and three-dimensional imaging of posterior ocular
structures.
2.1.2 Indications for Use
The PLEX® Elite 9000 is a non-contact, high resolution, wide field
of view tomographic and biomicroscopic imaging device intended
for in-vivo viewing, axial cross-sectional and three-dimensional
imaging of posterior ocular structures. The device is indicated for
visualizing posterior ocular structures including, but not limited to,
retina, retinal nerve fiber layer, ganglion cell plus inner plexiform
layer, macula, optic nerve head, vitreous and choroid.
The PLEX® Elite 9000 angiography is indicated as an aid in the
visualization of vascular structures of the retina and choroid.
The PLEX® Elite 9000 is indicated as a diagnostic device to aid in
the detection and management of ocular diseases including, but
not limited to, macular holes, cystoid macular edema, diabetic
retinopathy, age-related macular degeneration and glaucoma.
2.1.3 Usage
The PLEX® Elite 9000 is designed for continuous use, although it is
expected that most sites operate the instrument for 10 hours or
less per day, indoors, within a medical office or hospital setting.
This setting shall have clean air, free of soot, vapors from
adhesives, grease or volatile organic chemicals. Other Operating
Environment specifications are given in Environmental Conditions
[}216]. Application–related warnings are given in this chapter and
elsewhere.
The PLEX® Elite 9000 is not a portable device. It is intended for
placement in one location. However, there is no permanently
installed infrastructure associated with the instrument, and it can
be moved between locations following the applicable guidelines
and warnings in this User Manual and particularly in Safety and
Certifications [}9].
 Loading...
Loading...