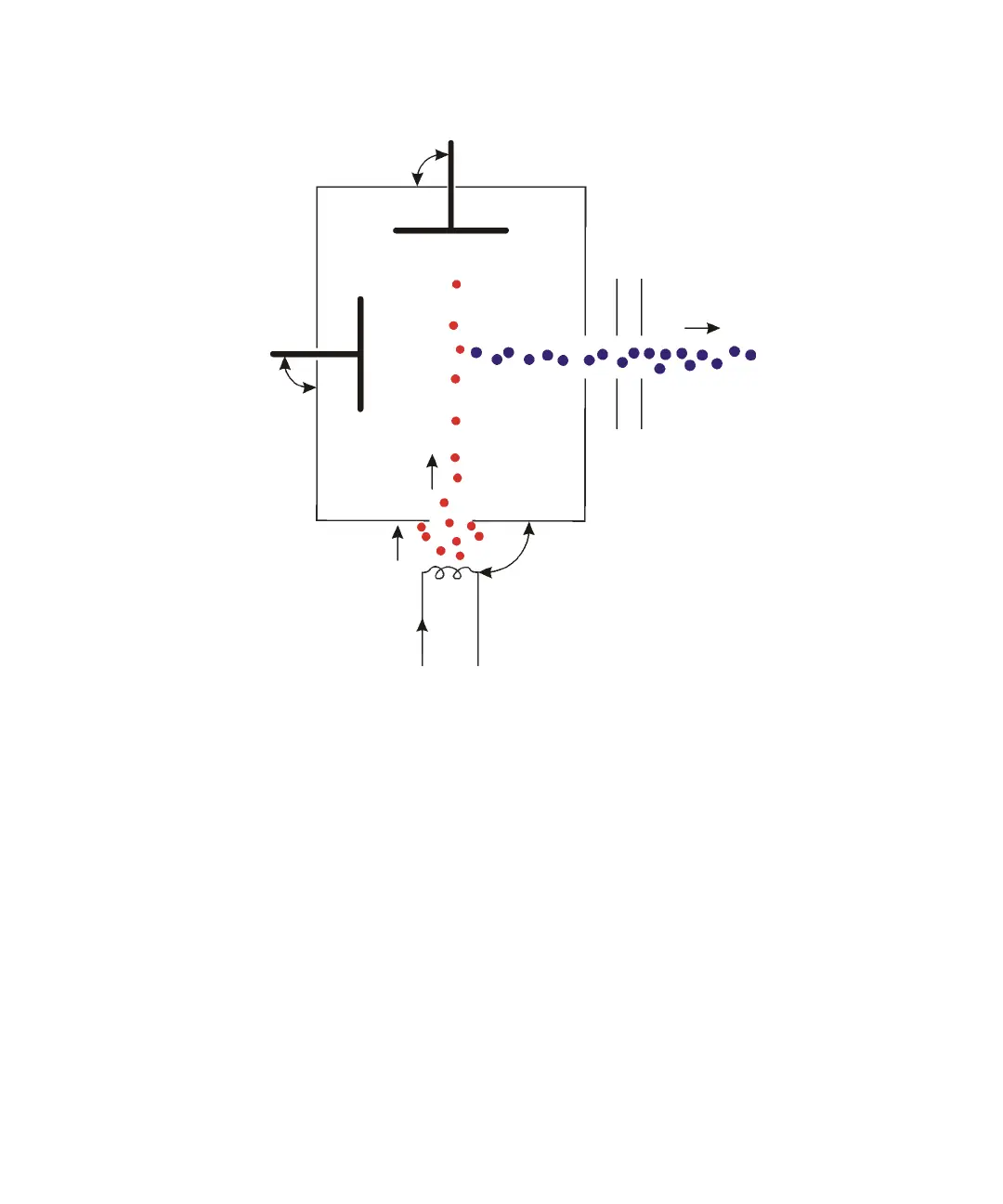
G-4 Theory and Principles of Operation
Electron impact ionization:
Chemical ionization
In contrast to EI (where the vaporized sample molecules are ionized by
colliding with high energy electrons from the filament), chemical ionization
(CI) uses a relatively high-pressure reagent gas (the CI gas) in the ion source
to moderate the collision energy. Ammonia, iso-butane, and methane are
commonly used as CI gases.
Electron bombardment ionizes the CI gas molecules within the ion source. The
sample molecules are then ionized in reactions with ionized CI gas molecules.
To achieve the relatively high pressures required within the ion source ion
chamber, while maintaining an adequate vacuum elsewhere, the chamber is
partially gas-tight.
Unlike the EI source, the CI inner source does not include an electron trap,
since insufficient electrons are likely to reach an electron trap under CI
Electron trap
Trap voltage
Ion repeller
epeller voltage
Ion chamber
Emission current
Electron energy (volts)
Emission
current
Filament
Filament current
Ion current
 Loading...
Loading...







