Milestone Srl MA245-001 – ultraWAVE 3 – User Manual
112
11 21CFR – PART 11 SOFTWARE
The U.S. FDA Code of Federal Regulations (CFR) Title 21 Part 11 (
Elettronic records: elettronic
signatures
) defines the criteria under which electronic records and electronic signatures are considered
trustworthy, reliable, and equivalent to paper records. The EasyCONTROL software is fully in compliance
with this regulation.
Thought the settings of the software it is possible to switch on all the functions requires by the 21CFR-11
regulation.
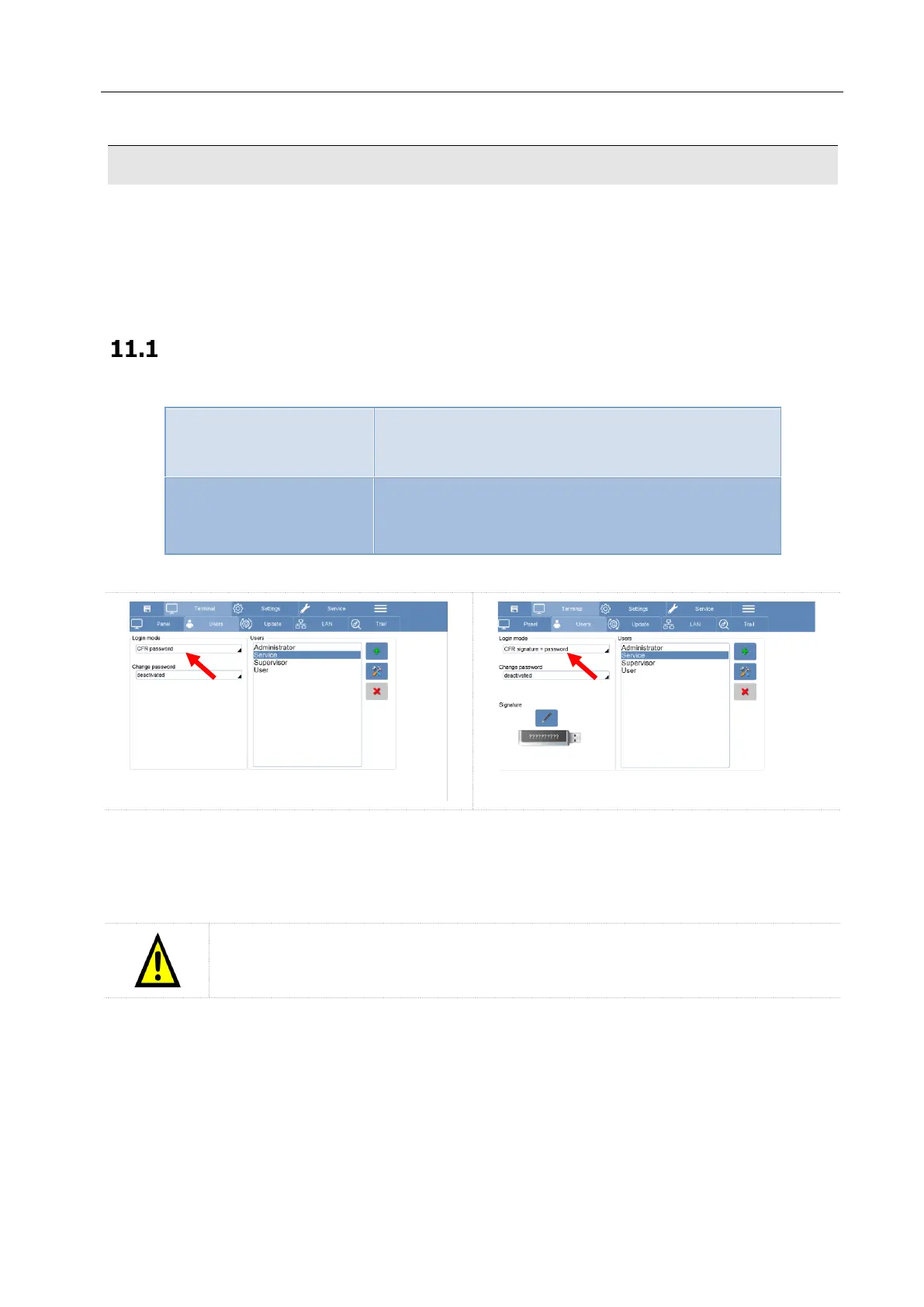
Activation of the 21CFR – part 11
The Login mode in accordance with the 21CFR-11, can be done in two different ways
Create new user profiles with different access privileges.
Each profile have a specific password to login.
Create new user profiles with different access privileges.
Each profile have a specific password with a signed USB
key, both necessary for the login.
You can set a password expiration. At the end of its lifetime, the software will ask to change the old
password with a new one.
Activating this function, the software immediately asks to change the old password with a new one.
The password expiration is not valid for the Service user profile.
Immediately after the activation of the CFR Signature + password, the local service
engineer must create his own signed USB with access to the service profile.

 Loading...
Loading...