Chapter 1—Introduction
10 BD Alaris
TM
System with Guardrails
TM
Suite MX User Manual
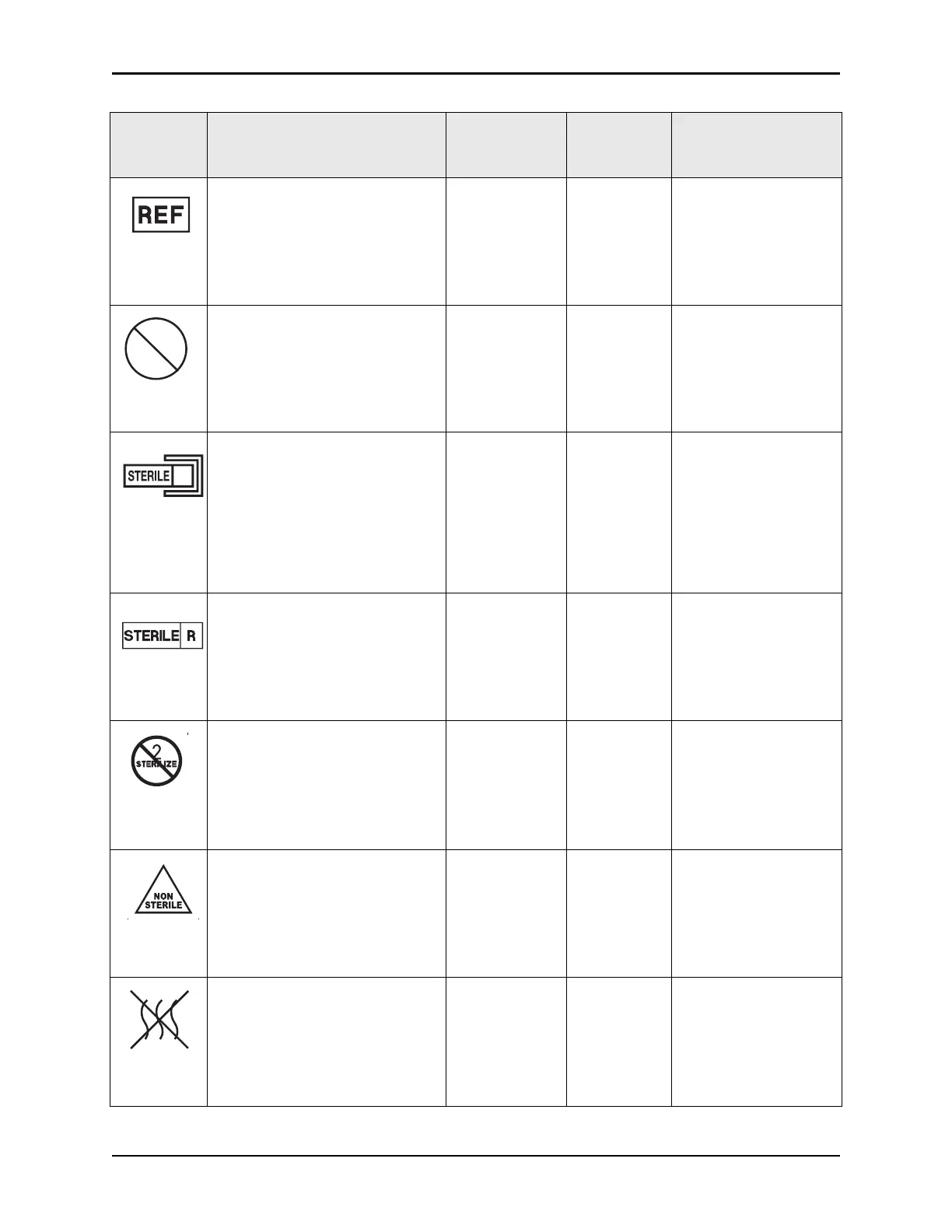
Indicates the manufacturer's
catalogue number so that the medical
device can be identified.
Catalogue
number
Disposable
packaging
Ref 5.1.6 ISO 15223-1
Medical Devices-
Symbols to be used with
medical device labels,
labeling and information
to be supplied-Part 1:
General Requirement
Indicates a medical device that is
intended for one
use, or for use on a
single patient during a single
procedure.
Do not re-use Disposable
packaging
Ref 5.4.2 ISO 15223-1
Medical Devices-
Symbols to be used with
medical device labels,
labeling and information
to be supplied-Part 1:
General Requirement
Indicates the presence of a sterile
fluid path w
ithin the medical device
in cases when other parts of the
medical device, including the
exterior, might not be supplied
sterile.
The method of sterilization shall be
indicated in
the empty box, as
appropriate.
Sterile fluid path Disposable
packaging
Ref 5.2.3
ISO 15223-1
Medical Devices-
Symbols to be used with
medical device labels,
labeling and information
to be supplied.
Indicates a medical device that has
been sterilized using irradiation.
S
terilized using
irradiation
Disposable
packaging
Ref 5.2.4 ISO 15223-1
Medical Devices-
Symbols to be used with
medical device labels,
labeling and information
to be supplied-Part 1:
General Requirement
Indicates a medical device that is not
to be resterilized.
Do
not resterilize Disposable
packaging
Ref 5.2.6 ISO 15223-1
Medical Devices-
Symbols to be used with
medic
al device labels,
labeling and information
to be supplied-Part 1:
General Requirement
Indicates a medical device that has
not been subjected to a sterilization
process.
Non-sterile Disposable
pac
kaging
Ref 5.2.7 ISO 15223-1
Medical Devices-
Symbols to be used with
medical device labels,
labeling and information
to be supplied-Part 1:
General Requirement
Indicates a medical device that is
non-pyrogenic.
Non-pyrogenic Disposable
packaging
Ref 5.6.3
ISO 15223-1
Medical Devices-
Symbols to be used with
medical device labels,
labeling and information
to be supplied-Part 1:
General Requirement
Symbol
(Graphical
Image)
Definition Symbol Title Symbol
Location
Ref. No. Standard
 Loading...
Loading...